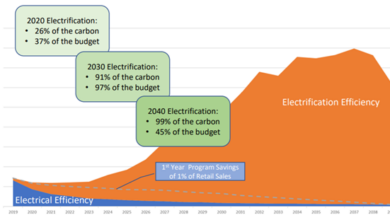
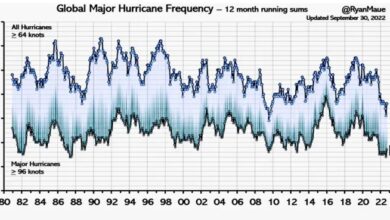
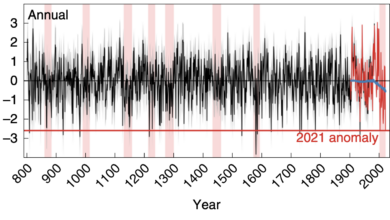
The Fundamentals of Ocean pH – Raised by that?

R. Cohen and W. Happer – September 18, 2015 (republished November 21, 2021)
1. Introduction
It is commonly known that the CO2 released from the burning of fossil fuels causes very dangerous acidification of the oceans. In fact, the oceans will remain alkaline and habitable for the foreseeable future. This brief note is a quantitative assessment of the physical chemistry of ocean pH. High school chemistry and algebra should provide enough background to follow the discussion. An excellent introduction to the chemistry of the oceans can be found in the book: Seawater: Its Composition, Properties and Behavior, by Wright and Colling. You can find more detailed information on ocean pH in Tans’s recent review.
Ocean alkalinity without buffer
Oceanic water is salty due to the weathering of the earth’s rocks by rainwater, and from dissolved salts from the ocean floor, especially near plate-spread boundaries. Salt water contains positive ions (cations), mainly sodium Na +, magnesium Mg2 +, calcium Ca2 + and potassium K +. To be more clear, we can think that the ions come from the strong base NaOH, and the potassium hydroxide, KOH (lye) and less soluble magnesium hydroxide, Mg(OH) 2 and calcium hydroxide Ca(OH) 2 (lime slaked). The ocean also contains negative ions (anions), mainly chloride Cl− and sulfate SO2, which we can consider anions to come from the strong acids, hydrochloric acid, HCl and sulfuric acid, H2SO4. For a better word, we will refer to cations of strong bases and anions of strong acids as pH-independent ions. The concentration of pH-independent ions is not affected by normal changes in the pH of the water.
Download the full article here: 2015 Cohen Happer Ocean pH Fundamentals

Roger Cohen is a co-founder of the CO2 Alliance and a respected physicist with major contributions to materials science and industrial management. He died on September 10, 2016, less than a year after completing this important paper.