CDC advisors support broader use of booster COVID vaccine: Shots
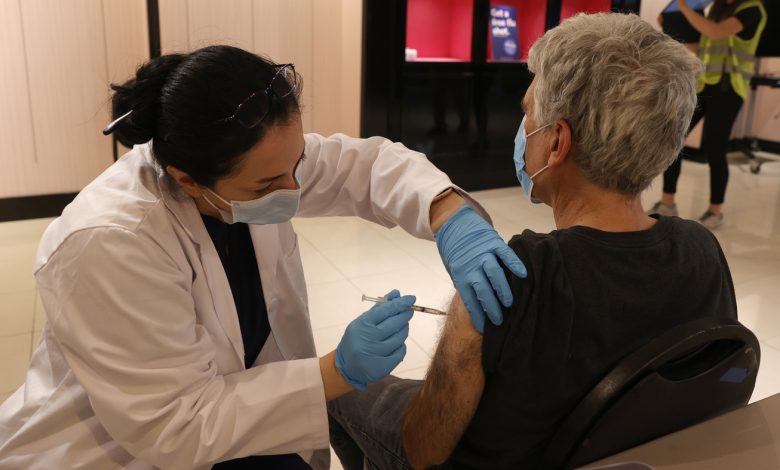

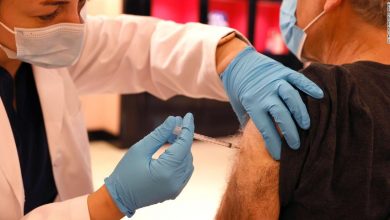
Safeway pharmacist Shahrzad Khoobyari injected a Pfizer-BioNTech COVID-19 booster into Norman Solomon’s arm in San Rafael, California, in October.
Justin Sullivan / Getty Images
hide captions
switch captions
Justin Sullivan / Getty Images
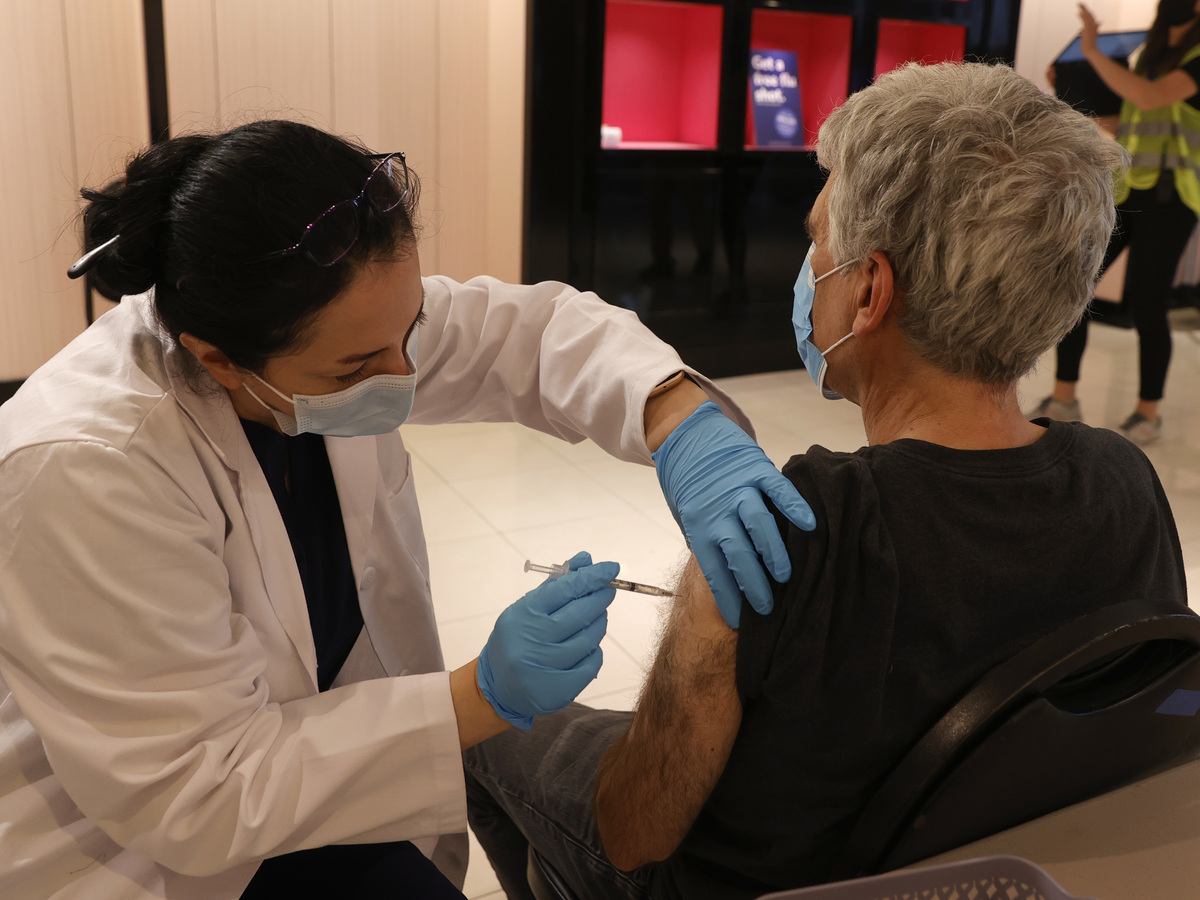
Safeway pharmacist Shahrzad Khoobyari injected a Pfizer-BioNTech COVID-19 booster into Norman Solomon’s arm in San Rafael, California, in October.
Justin Sullivan / Getty Images
A group of experts advising the Centers for Disease Control and Prevention has advocated expanding the Moderna and Pfizer-BioNTech COVID-19 vaccines to all adults.
The experts met on Friday afternoon just hours after the Food and Drug Administration Authorize boosters for people 18 years of age and older.
The Advisory Committee on Immunization Practices voted in favor of a change to the COVID-19 vaccination policy saying that people 50 years of age and older should get a booster shot if they have been vaccinated with the mRNA vaccine (Moderna or Pfizer) at least six months prior. The recommendation also applies to people 18 years of age and older in long-term care facilities.
For those aged 18 and under, under 50, the council backed a policy that said they could receive a boost based on individual risks and benefits.
An analysis from a CDC working group concluded that the balance of benefits and risks for enhancement is most pronounced for older adults. The team also notes that the latest data on myocarditis, an inflammation of the heart that is rare after vaccination but often occurs in young men, is “consoling so far.”
The next step is for CDC Director Rochelle Walensky to issue a statement on the committee’s recommendations and issue a formal position from the public health authority.
The director usually follows the recommendations of the board but She approved aspects of the committee’s September decision on Pfizer’s first in a rare departure.